A 49% lower risk of melanoma coming back after surgery sounds like hype copy, but it’s a real number from Moderna and Merck’s individualized mRNA vaccine plus Keytruda combo at 5‑year follow‑up. That’s what mRNA cancer vaccines quietly did in a Phase 2b trial while Reddit argued about “curing cancer.”
2026 is when this stops being a handful of glamorous proofs‑of‑concept and becomes a practical question: can we actually run this at scale, pay for it, and plug it into messy real‑world oncology?
TL;DR
- 2026 Phase‑3 data won’t “cure cancer,” but they will likely prove mRNA cancer vaccines work as adjuvant/combination tools that cut recurrence in specific tumors.
- The hard part shifts from “does the biology work?” to “can we manufacture a personalized neoantigen vaccine in weeks, not months, and at a price payers tolerate?”
- If the readouts are positive, the real disruption is economic and logistical: oncology becomes part‑software pipeline, part‑biotech factory, not just more chemo plus a new drug name.
Why 2026 Matters for mRNA Cancer Vaccines
On paper, BioNTech calls 2026 a “catalyst‑rich year” with around 15 Phase‑3 oncology trials running and seven late‑stage readouts on deck. In practice, it’s a referendum on whether mRNA cancer vaccines graduate from “clever science fair” to “new standard‑of‑care component.”
You already saw the pandemic story: same mRNA platform, different sequence, shipped at software speed. Oncology is the adult‑mode version of that story.
Now the test is harder:
- FixVac programs like BNT113 (HPV16+ head‑and‑neck cancer) are “off‑the‑shelf” mRNA cocktails aimed at shared tumor antigens.
- iNeST programs like BNT122 (autogene cevumeran) are fully personalized neoantigen vaccines built from each patient’s tumor mutations.
- Moderna/Merck’s intismeran autogene (formerly mRNA‑4157) is the other big neoantigen player, already showing multi‑year recurrence‑free survival gains in resected high‑risk melanoma.
If those Phase‑3 readouts are clean and positive, the argument “mRNA works in infectious disease but maybe not in cancer” dies overnight. The question flips from can the immune system be taught? to can the healthcare system keep up?
That’s the pivot.
What the Phase‑3 Readouts Are Actually Testing
The headline fantasy is “we finally cure cancer.” The actual endpoints are way more boring and way more important.
Most of these mRNA cancer vaccines are being tested in adjuvant or combination settings:
- Adjuvant = after tumor removal, to prevent recurrence.
- Combination = layered on top of checkpoint inhibitors like Keytruda or cemiplimab, not replacing them.
The Moderna/Merck melanoma study is the template. Patients had high‑risk Stage III/IV melanoma completely resected, then either:
- Keytruda alone, or
- Keytruda + individualized mRNA vaccine encoding up to a few dozen of that patient’s tumor‑specific neoantigens.
At five years, the combo arm still shows a roughly 49% reduction in recurrence risk versus Keytruda alone. That’s not “no cancer ever again.” It’s “your odds get tilted meaningfully in your favor.”
BioNTech’s Phase‑3 bets are set up similarly:
- BNT113 in first‑line HPV16+, PD‑L1+ head‑and‑neck cancer.
- BNT122 in adjuvant, circulating‑tumor‑DNA‑positive colorectal cancer.
- Other FixVac and iNeST trials layered on top of immunotherapy backbones.
Endpoints that matter:
- Recurrence‑free survival (RFS)-does the cancer come back later or less often?
- Progression‑free survival (PFS)-in metastatic settings, does the disease stay stable longer?
- Overall survival (OS)-the hardest, longest‑to‑mature endpoint, and the one regulators and payers care about most.
If these Phase‑3s hit their RFS/PFS marks and show a believable OS trend, they don’t prove “cure.” They prove something more actionable: adding an mRNA cancer vaccine to existing care is worth the cost and complexity for certain tumor types.
For you as a patient or payer, that’s the difference between another press release and a new line item in the treatment protocol.
The Real Bottlenecks: Manufacturing, Biology, Regulation, Cost
The weird thing about mRNA cancer vaccines is that biology might be the least scary part now.
Peer‑reviewed reviews in 2025-2026 are surprisingly bullish on the core mechanism: mRNA is good at provoking neoantigen‑specific T‑cell responses. The problems are everything around that mechanism.
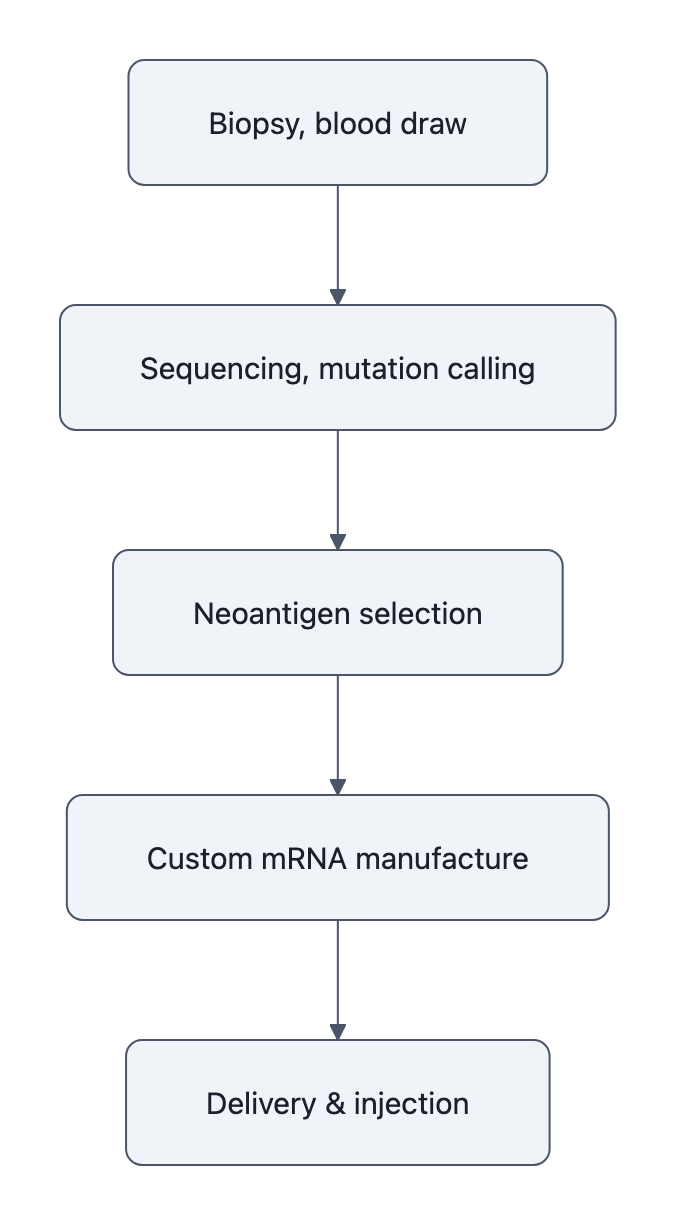
1. Manufacturing personalized vaccines at speed
A personalized neoantigen vaccine like BNT122 or intismeran is a pipeline, not a pill:
- Biopsy tumor and blood.
- Sequence DNA/RNA, call mutations, predict candidate neoantigens.
- Run proprietary algorithms to pick the best set.
- Manufacture a custom mRNA formulation.
- Ship and inject on a timetable that still matters for minimal residual disease.
Every one of those steps can slip. Each step adds cost. MDPI’s 2025 review is blunt: individualized RNA vaccines are logistically complex and expensive, with timelines measured in many weeks.
And unlike a COVID shot, losing three months in the adjuvant window can be the whole ballgame.
2. Tumor microenvironment and biology that doesn’t care about your press release
The Nature npj Vaccines review on prostate cancer makes the key point: you can induce gorgeous T‑cell responses on paper and still lose in the tumor microenvironment.
Solid tumors are not sitting ducks. They’re:
- Hypoxic, acidic, and immunosuppressive.
- Heterogeneous, your chosen neoantigens may not be on every clone.
- Guarded by regulatory cells and nasty cytokine gradients that shut T‑cells down.
That’s why so many designs are “mRNA vaccine + checkpoint inhibitor + maybe something else.” The vaccine teaches the immune system the target; the other drugs break the tumor’s cloaking and suppression. Phase‑3 readouts will show whether that combined stack consistently moves meters, not millimeters.
3. Regulation and economics: who pays for a bespoke biologic?
Even if the Phase‑3s succeed, the 2025 Cancers review estimates first large‑scale commercial approvals for RNA cancer vaccines closer to the late 2020s. One reason: health systems have to decide what “worth it” means.
Think about pricing:
- A fully personalized mRNA cancer vaccine could easily start in the high five to six figures per course.
- It’s on top of surgery, imaging, and a $150k+ checkpoint inhibitor.
Regulators will want strong survival data or at least very clear, durable recurrence‑free survival gains. Payers will push hard on biomarkers: which patients actually benefit enough to justify the cheque?
The medicine might be precision. The reimbursement won’t be.
If Readouts Are Positive: How Cancer Care Would Change
Assume the semi‑optimistic scenario: 2026 readouts from BioNTech’s FixVac/iNeST and Moderna/Merck’s follow‑ups are clearly positive, and regulators start giving green lights over the next 3-5 years.
That’s not “we cured cancer.” It is a workflow revolution.
Oncology becomes a software‑plus‑biomanufacturing problem
Today, your path after tumor resection is mostly:
- Watchful waiting + scans
- +/- chemo, radiation, immunotherapy based on risk
In an mRNA‑positive world, an early‑stage high‑risk patient journey looks more like:
- Resection.
- Immediate tumor sequencing and neoantigen pipeline spin‑up.
- Tumor board decides: does this patient qualify for a vaccine add‑on?
- A few weeks later, infusions or injections of a personalized mRNA cancer vaccine alongside a checkpoint inhibitor.
Clinics will need:
- On‑site or regional sequencing capacity and robust bioinformatics.
- Interfaces to manufacturer pipelines (ordering, tracking, QA).
- New scheduling and toxicity‑management protocols for high‑dose, repeat mRNA in oncology.
You don’t just add “BNT122” to a dropdown. You refactor how you run a cancer center.
And you can see why AI and automation suddenly matter here: triaging scans, prioritizing sequencing queues, optimizing manufacturing runs. This is as much an operations research problem as a pharmacology one.
Economics: adjuvant recurrence reduction is where the money is
From a health‑system point of view, adjuvant vaccines that reduce recurrence are the dream use‑case:
- Recurrent metastatic disease is where costs explode.
- Avoiding a handful of very expensive relapses can pay for a lot of one‑time adjuvant therapy.
So if Phase‑3 results show, say, a 30-50% relative reduction in recurrence for clearly defined high‑risk patients, expect:
- Rapid guideline uptake in wealthy systems for those niches (e.g., ctDNA+ colorectal, high‑risk melanoma, biomarker‑defined head‑and‑neck).
- Aggressive negotiation on price, plus value‑based contracts (“pay more if recurrence really drops as promised”).
- Inequality in access: large academic centers adopt early; community hospitals and low‑income countries lag years behind.
The economic story here isn’t “cure cancer and save trillions.” It’s “spend more upfront to avoid the really ruinous cases later” and then watch oncology budgets and workflows reorganize around neoantigen pipelines.
Expect a long “mRNA oncology decade,” not a 2026 miracle
Positive readouts in 2026 flip the default from “skeptical” to “assume this works and iterate.” That sets up:
- A proliferation of tumor‑specific vaccines (lung, prostate, GI, etc.).
- Faster design cycles for combinations as we get better biomarkers and TME‑modifying agents.
- Steady expansion from niche high‑risk groups to broader indications, if cost and manufacturing curve down.
If you’re looking for a date when cancer ends, you’ll be disappointed. If you’re watching for when oncology starts behaving like a data‑heavy, pipeline‑driven industry instead of pure one‑drug‑at‑a‑time pharmacology, circle 2026.
It’s not the epilogue. It’s chapter one of a new genre.
Key Takeaways
- 2026 Phase‑3 readouts for mRNA cancer vaccines will likely validate them as effective adjuvant/combination tools that reduce recurrence in specific cancers, not universal cures.
- The endpoints that matter are recurrence‑free and progression‑free survival, plus believable trends in overall survival, not “no more cancer ever.”
- The biggest constraints after positive data are manufacturing pipelines, tumor microenvironment biology, regulatory proof thresholds, and the raw cost of personalized neoantigen vaccines.
- If approved, these therapies will turn cancer care into a sequencing‑plus‑software‑plus‑biomanufacturing problem, reshaping oncology workflows and budgets over the next 3-5 years.
- For technically curious generalists, the real story is not curing cancer but how these vaccines will quietly rewrite the economics and logistics of cancer care.
Further Reading
- BioNTech Provides Strategic Business Update and Outlines 2026 Areas of Focus, BioNTech’s own roadmap for its oncology Phase‑3 programs and expected 2026 readouts.
- Moderna, Merck announce 5‑year data for intismeran + KEYTRUDA, Company release detailing sustained recurrence‑free survival benefit in melanoma.
- Current Progress and Future Perspectives of RNA‑Based Cancer Vaccines: A 2025 Update, Peer‑reviewed overview of mRNA cancer vaccine trials, mechanisms, and practical bottlenecks.
- Breaking barriers in prostate cancer: the mRNA vaccine breakthrough and what comes next, npj Vaccines review on immune responses, tumor microenvironment limits, and combination strategies.
- mRNA cancer vaccine shows protection at 5‑year follow‑up, Moderna and Merck say, Independent coverage putting the melanoma vaccine data in context and stressing the need for Phase‑3 confirmation.
mRNA cancer vaccines won’t give you a tidy “we cured cancer” headline in 2026. They’re more likely to do something stranger and ultimately more consequential: turn cancer care into a pipeline problem that looks a lot more like software and a lot less like chemistry.

